INNO-LiPA® HPV Genotyping Extra II for use on self-collected first-void urine samples
HPV (Human Papilloma Virus) is a sexually transmitted virus. Certain genotypes of this virus are known to cause cancers in both men and women, but most notably, HPV is the primary cause of cervical cancer in women1,2,3.
When HPV infection is diagnosed and genotyped effectively, the potential for this virus to cause cancer can be substantially reduced. 2,3
Historically, testing for this virus has required the collection of tissue samples using cervical or other body-site swabs. The sampling process can be embarrassing and inconvenient, and statistics show that for a variety of cultural, religious, and lifestyle reasons, women are not undergoing cervical cancer screening as often as they should.4,5
By making it possible to genotype HPV infections with a urine sample that is painlessly collected by the patient in privacy, it is hoped that HPV testing rates can be increased, vaccine efficacy studies simplified, and lives saved.6,7,8
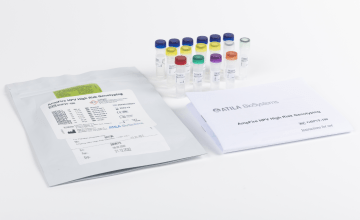
The already widely used genotyping assay for HPV detection and specification from Fujirebio, INNO-LiPA HPV Genotyping Extra II, has now received a CE Mark for use with self-collected UCM-preserved first-void urine samples.
The article continues below.
Why consider INNO-LiPA HPV Genotyping Extra II for testing of self-collected samples?
INNO-LiPA HPV Genotyping Extra II – high sensitivity and specificity on cervical sample & urine
- Sensitive and specific identification of 32 genotypes of HPV using cervical swab samples (liquid-based cytology)9 and paraffin-embedded tissue (FFPE)10
- SPF10 PCR target used can genotype infections even in case of degraded DNA (FFPE samples) and in case of low viral load, “masked” HPV types, and mixed HPV infections.9,10,11
- Newly validated urine protocol makes it now possible to achieve sensitive & specific genotyping results also on UCM-preserved first void urine samples (see IFU)
In order to collect urine samples standardized and reliably, Fujirebio Europe also distributes the CE marked Colli-Pee™ first-void urine collection device manufactured by Novosanis. This patented urine collection device simplifies the private and painless collection and preservation of first-void urine samples from both men and women.
References
-
Walboomers JM et al. Human papillomavirus is a necessary cause of invasive cervical cancer worldwide. J Pathol 1999;189:12–19
-
IARC Monogr Eval Carcinog Risks Hum 2012;100B:255–313
-
F. Xavier Bosch et al. Comprehensive Control of Human Papillomavirus Infections and Related Diseases. Vaccine. 2013 Nov 22; 31(0 8): I1–31
-
Verdoodt et al. 2015. Reaching women who no not participate in the regular cervical cancer screening programme by offering self-sampling kits: a systematic review and meta-analysis of randomized trials. Eur J Cancer. 51 (16):2375-85
-
Racey CS et al. Self-collected HPV Testing Improves Participation in Cervical Cancer Screening: A Systematic Review and Meta-analysis. Can J Public Health 2013;104(2):e159-e166
-
O'Leary MC et al. HPV type-specific prevalence using a urine assay in unvaccinated male and female 11- to 18-year olds in Scotland. Br J Cancer 2011. 104(7):1221-1226.
-
Ducancelle A. et al. Interest of human papillomavirus DNA quantification and genotyping in paired cervical and urine samples to detect cervical lesions. Arch Gynecol Obstet 2014; 290(2):299-308
-
Burroni E. et al. Human papillomavirus prevalence in paired urine and cervical samples in women invited for cervical cancer screening. J Med Virol. 2015; 87(3):508-15
-
Xu L. et al. Clinical Evaluation of INNO-LiPA HPV Genotyping Extra II Assay Using the VALGENT Framework. Int J Mol Sci. 2018; 19(9), 2704
-
Ahmadi S. et al. Human Papilloma Virus Genotype Distribution in Cervical lesions in Zanjan, Iran. Asian Pac J Cancer Prev. 2017; 18(12):3373-3377
-
Sohrabi et al. Is incidence of multiple HPV genotypes rising in genital infections? J Infect Public Health. 2017; 10(6):730-733