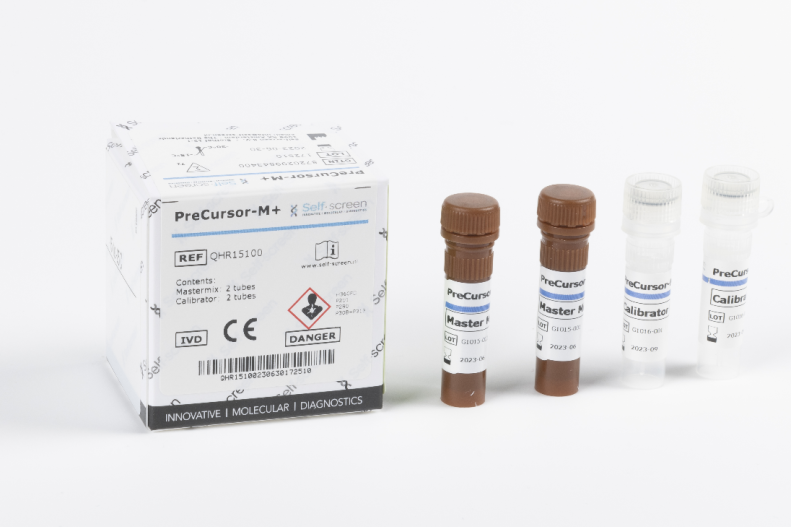
PreCursor-M+
Qualitative detection of promotor hypermethylation of 2 cervical cancer associated genes (FAM19A4 and hsa-miR124-2).
Identify HPV positive women with high short-term progression to (pre)cancer.
Special Features:
- Sample types:
- Bisulfite-converted DNA isolated from physician collected cervical specimens or self-collected vaginal specimens
- Clinical performance - Long-term data:
- Most evaluated and validated methylation assay to identify HPV positive women with progressing cervical disease in direct need for colposcopy or other follow-up procedures1,2
- Demonstrating very high sensitivity for cervical cancer detection (95.0-100%) in HPV-positive cervical specimens2,3,4
- Longitudinal data: The long-term risk for cancer with a negative PreCursor-M+ is lower than with a negative cytology result; for CIN3 the long-term risk is similar as cytology5,6
- Flexible:
- Suitable for various sample types, including self-samples and common Liquid Based Cytology media types
- Compatible with standard DNA extraction methods
- Reliable:
- Build-in check for sample quality ensuring reliable results
Precursor-M+ is a registered trademark of the legal manufacturer, Self-screen B.V., the Netherlands, and is distributed by Fujirebio Europe.
PreCursor-M+
Product number 81352
72 Tests
CE marked
Please contact your local Fujirebio representative for the availability of this product in your country.