INNO-LiPA® HPV Genotyping Extra II
Line probe assay, based on the reverse hybridization principle, designed for the identification of 32 different genotypes of the human papillomavirus (HPV).
- Individual identification of 32 genotypes.
- One genotype = one probe line.
- Ready-to-use mastermix.
- Automation.
- Primer mix for RT-PCR application.
- Cervical cells, first-void urine and formalin-fixed paraffin-embedded tissue (CE).
Product number 81534
Product number 81533
Click here to navigate
- Contact sales for information
- Details
- Conditions of sale
- Citations
- Documentation
- Insights
- Product inquiry
- Related products
- Webinars
-
Details
Features and benefitsFull genotyping
- Simultaneous detection of 32 HPV genotypes.
- SPF10 Plus provides high test sensitivity due to the precision of the short 65-base pair PCR product, even in case of degraded DNA (FFPE samples), low viral load and mixed HPV infection.1,2,3
- SPF10 Plus permits simultaneous detection of multiple genotypes in a single sample.
Built-in controls
- Special HPV control lines to confirm and detect the presence of a broad range of mucosal HPV genotypes plus additional controls to monitor sample processing (hDNA line, conjugate control line).
Streamlining
- Easy and ready-to-use master mix, with Taq DNA polymerase provided for minimum manipulation steps and hands-on time.
CE Applications:
- Cervical cells collected in Surepath medium or PreservCyt medium.
- First-void urine with preservative.
- Formalin-fixed paraffin embedded (FFPE) material.
RUO Applications:
- HPV Primer Mix SPF10 Plus for Real-Time PCR application.
Automation
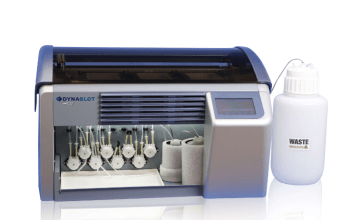
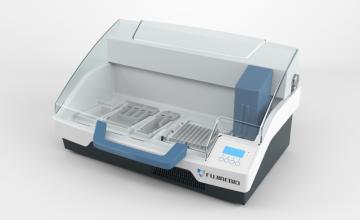
- Auto-LiPA™ 48: up to 48 tests per run from sample incubation to color development.
- AutoBlot 3000H: up to 20 tests per run from sample incubation to color development.
- TENDIGO®: up to 10 tests per run from sample incubation to color development.
- Results within +/- 3 hours.
- Minimum hands-on time, accurate results.
- Interpretation software for complete traceability.
References- Xu L. et al. Clinical Evaluation of INNO-LiPA HPV Genotyping Extra II Assay Using the VALGENT Framework. Int J Mol Sci. 2018; 19(9), 2704
- Ahmadi S. et al. Human Papilloma Virus Genotype Distribution in Cervical lesions in Zanjan, Iran. Asian Pac J Cancer Prev. 2017; 18(12):3373-3377
- Sohrabi et al. Is incidence of multiple HPV genotypes rising in genital infections? J Infect Public Health. 2017; 10(6):730-733
-
Conditions of sale
To read the end user conditions of sale for this product please visit our Resource center.
-
Citations
The BIOZ badges associated with Fujirebio products include peer-reviewed citations derived from scientific studies using Fujirebio products. Please note that the peer-reviewed citations do not reflect the regulatory status of Fujirebio products. Users should refer to the specific product documentation and any (clinical) claims made therein in order to ensure compliant use. For each country or geographic region, users must verify the related regulatory status of the Fujirebio product.
-
Documentation
Browse regulatory documents for this product
Create a (free!) eServices account and start browsing all product documentation right away.
Get access to this section and more
Create a free eServices account now and instantly access multiple digital resources:
- Product documentation
- Selected scientific posters
- Product leaflets
- How-to videos and more…
-
Insights
Mar 10, 2026Advancing anal cancer prevention: Screening expansion and the emerging role of host-cell DNA methylation
Anal cancer incidence remains low in the general population but is significantly elevated in defined at-risk groups, including people living with HIV...
Feb 4, 2026HPV genotyping in FFPE tissue: meeting the challenges of archival sample analysis
Human papillomavirus (HPV) is a necessary cause of cervical cancer and is increasingly recognized as an important etiological factor in a range of...
Sep 24, 2025Clinical validation of the PreCursor-M AnoGYN test: Tailoring treatment of anal and vulvar cancer precursors
A major challenge for the correct implementation of the anal cancer screening guidelines is the generally limited availability of High-Resolution...
-
Product inquiry
-
Related products