Aβ deposition and clearance: a key feature of the ageing brain
In this article series we aim at highlighting the current state of knowledge and the latest developments in the field of Alzheimer’s disease (AD) testing. This chapter looks closer at Aβ deposition and clearance as key feature of the ageing brain.
Other articles in this series:
- Altered proteins in brain neurodegenerative diseases
- Using CSF biomarkers to link pathology and clinical presentation
- How to perform a lumbar puncture
- Handling and transportation of CSF samples
- CSF biochemical pattern interpretation
- New criteria for Alzheimer’s disease
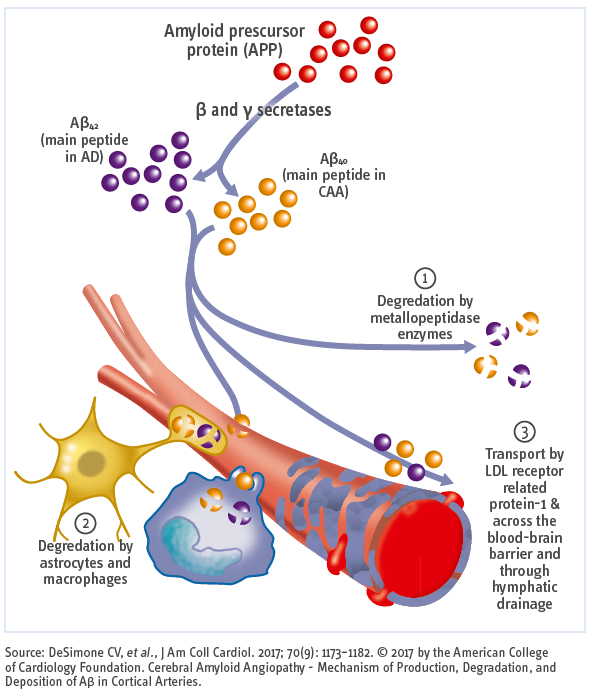
Aβ40 is the most abundant amyloid protein and less neurotoxic than Aβ42, which is less abundant, highly insoluble, severely neurotoxic, and more aggregation-prone.
Amyloid peptides can be degraded by metalloproteinases, astrocytes, or macrophages, or are transported across the blood–brain barrier via low-density lipoprotein (LDL) receptor–related protein-1 and drained via the lymphatic system.
The pathogenic pathways of cerebral amyloid angiopathy (CAA) and AD intersect at the levels of Aβ generation, its circulation within the interstitial fluid and perivascular drainage pathways and its brain clearance:
- Aβ40: Primarily associated with vascular Aβ deposition
- Aβ42: Primarily associated with plaque Aβ deposition
Vascular deposits also contain Aβ42, but the proportion of Aβ40 is higher than that found in plaques. Impairment of perivascular drainage induces a self-reinforcing cycle of Aβ deposition, loss of vascular smooth muscle cells and vasoactivity, and further reduction in clearance altogether causing CAA.
The AD and CAA pathways seem to diverge with respect to how they cause tissue injury: AD pathology promotes neuronal and synaptic loss whereas CAA generates focal tissue lesions via haemorrhagic and ischaemic vascular brain injury.
CAA occurs in 85% to 95% of patients with AD, has a signifi cant impact on vessel health, and is an important contributor to cerebrovascular pathology in AD.
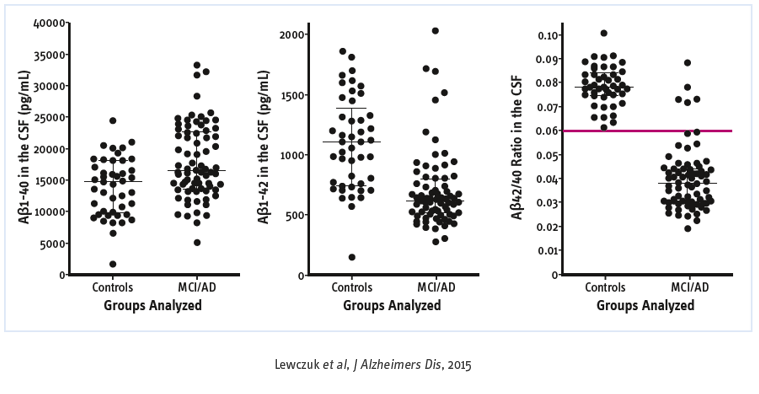
Added value of the Aβ42/40 ratio
Aβ peptide is produced from a transmembrane Aβ precursor protein, sequentially cleaved by β- and γ-secretase. Cleavage of APP by γ-secretase generates a number of Aβ isoforms. Aβ42, a 42 amino acid-long peptide, has the highest propensity for aggregation and appears to be the predominant species in neuritic plaques. Although the concentration of Aβ40, has been reported to be unaltered in AD, the Aβ42/40 ratio has been suggested to be superior to the concentration of Aβ42 alone in discriminating patients with AD.
In a population of normal subjects and AD patients, the distribution of total Aβ (40 and 42) follows a Gaussian distribution for both normal subjects and AD patients, with Aβ40 making up about 70% of total Aβ. Further although many cases fall into the middle of the distribution with the majority having normally total Aβ, outliers are still present. Some AD patients will have high total Aβ (enhanced amyloidogenic processing of APP, also called “high producers”) and some cognitively normal subjects will have low total Aβ (reduced amyloidogenic) processing of APP, also called “low producers”.
This means that AD patients with a high total Aβ will show an incomplete CSF pattern and vice versa, normal subjects with low total Aβ will be classify as individual with sign of cerebral amyloidosis. In all three cases (normal, low and high total Aβ) the ratio can correctly classify some doubtful CSF pattern. The ratio led to a reduction by half of the number of indeterminate profi les without changing the conclusion when usual biomarkers (Aβ42 and P-tau) were concordant.
There is a consensus to recognize that the Aβ42/40 ratio is helpful to:
- Reflect interindividual difference in amyloidogenic-APP processing
- Solve undetermined core biomarker profiles of AD
- Decrease the impact of preanalytical and analytical sources of variability within and among centers.
Added value of Aβ40 alone
CAA diagnosis based on MRI findings alone is not clear. Amyloid imaging with amyloid-binding PET ligands can detect CAA, although they cannot discriminate vascular from parenchymal amyloid deposits. In addition, CSF markers may be useful, including levels of Aβ40 for CAA and anti-Aβ antibody for CAA-related inflammation (CAA-ri).
Recent findings indicate that the presence of one or more biomarkers plus one or more risk factors may be suggestive of CAA:
- Amyloid imaging with greater occipital uptake
- A decrease in CSF Aβ40 levels
Risk factors:
- General factors:
- Old age
- AD
- Genetic factors:
- CAA-related gene mutations in familial cases
- APOE in sporadic cases: ε4 as a risk factor for CAA
Impact of carrying the APOE4 allele
The brain Aβ pathology is inarguably associated with APOE ε4 status. Carrying the ε4 allele of the APOE gene encoding apolipoprotein E (APOE ε4) markedly increases the risk for AD and CAA. APOE ε4-mediated amyloid pathology depends on its neuronal LDL receptor–related protein 1 (LRP1). APOE ε4 decreases Aβ clearance without affecting Aβ production. According to the current concept, Aβ that accumulate in the brain in AD is likely due to its faulty clearance from the brain. LRP1 is a major efflux transporter for Aβ at the blood-brain barrier (BBB). Binding of Aβ to LRP1 at the abluminal side of the BBB initiates a rapid Aβ clearance from brain to blood via transcytosis across the BBB.
In summary, cognitive impairment in the ageing brain is typically driven by overlapping neurodegenerative and cerebrovascular pathologies. The impaired perivascular clearance of Aβ and the deficient neuronal LRP1 exacerbate the brain accumulation of Aβ peptides and subsequent deposition – the most likely cause of CAA and AD.
Bibliography
- APOE4-mediated amyloid-beta pathology depends on its neuronal receptor LRP1. Tachibana M, et al. J Clin Invest. 2019; 129(3): 1272-1277.
- Core cerebrospinal fluid biomarker profile in cerebral amyloid angiopathy: A meta-analysis. Charidimou A, et al. Neurology. 2018; 90(9): e754-e762. Review.
- Cerebral amyloid angiopathy and Alzheimer disease - one peptide, two pathways. Greenberg SM, et al. Nat Rev Neurol. 2020; 16(1): 30-42. Review.
- Cerebral amyloid angiopathy: emerging concepts. Yamada M. J Stroke. 2015; 17(1): 17-30. Review.
- Cerebrospinal Fluid Aβ42/40 Corresponds Better than Aβ42 to Amyloid PET in Alzheimer’s Disease. Lewczuk P, et al. J Alzheimers Dis. 2017; 55(2): 813-822.
- Cerebrospinal fluid amyloid-β 42/40 ratio in clinical setting of memory centers: a multicentric study. Dumurgier J, et al. Alzheimers Res Ther. 2015; 7(1): 30.
- Additional use of Aβ42/Aβ40 ratio with cerebrospinal fluid biomarkers P-tau and Aβ42 increases the level of evidence of Alzheimer’s disease pathophysiological process in routine practice. Sauvée M, et al. J Alzheimers Dis. 2014; 41(2): 377-386. Comparative Study.
- Amyloid beta peptide ratio 42/40 but not A beta 42 correlates with phospho- Tau in patients with low- and high-CSF A beta 40 load. Wiltfang J, et al. J Neurochem. 2007; 101(4): 1053-1059. Comparative Study.