Handling and transportation of CSF samples
In this article series we aim at highlighting the current state of knowledge and the latest developments in the field of Alzheimer’s disease (AD) testing. This chapter details the recommended procedure for handling and transportation of CSF samples.
Other articles in this series:
- Altered proteins in brain neurodegenerative diseases
- Using CSF biomarkers to link pathology and clinical presentation
- How to perform a lumbar puncture
- CSF biochemical pattern interpretation
- Aβ deposition and clearance: a key feature of ageing brain
- New criteria for Alzheimer’s disease
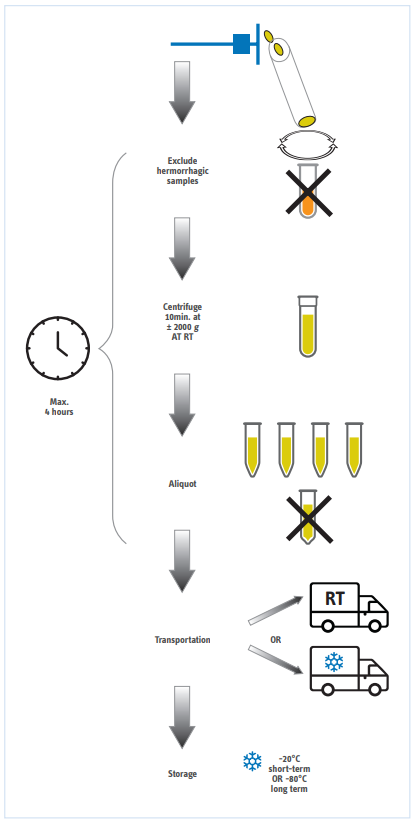
Cerebrospinal fluid (CSF) can be collected in the lumbar region by an experienced physician. Taking into account the necessary precautions (e.g, sterile environment) and other medical investigations (e.g, brain imaging), the lumbar puncture is generally accepted by the medical society as a safe procedure. Adsorption to plastic polymers can alter the biomarker concentrations and the extent of this adsorption differs between tubes. Therefore, always use the same polypropylene reference tube (within one lab).
Recommended procedure
- The CSF sample must be sent to the local laboratory without delay.
- » A CSF cell count is usually performed
- » The CSF sample is centrifuged in the original tube
- The CSF sample is aliquoted for basic and specific neurodegenerative disease biomarkers.
- » CSF should be aliquoted in tubes made of polypropylene
- » The volume needed for CSF analyses may vary between laboratories
- » The volume needed for Alzheimer specific biomarkers is between 0.5 mL and 1.0 mL
- Transportation: CSF samples can be sent by ordinary mail, at room temperature if the shipping time is less than two days. If the CSF sample is taken on a Friday, it can be frozen and sent to the central laboratory on dry ice the next week.
- Handling of CSF samples before analyses of AD biomarkers.
- » It is recommended to freeze CSF samples if the analysis cannot be performed within 48 hours. Samples sent frozen should be kept frozen until analysis.
- » CSF samples sent at room temperature should be frozen, and CSF samples sent frozen should be kept frozen.
Sample analysis on fresh samples upon arrival may result in slightly different CSF biomarker values compared to frozen samples but is unlikely to result in a different CSF biomarker profile.
Schematic representation
Bibliography
- The Alzheimer’s Association international guidelines for handling of cerebrospinal fluid for routine clinical measurements of amyloid β and tau. Hansson O, et al. Alzheimers Dement. 2021; 1-8.
- Measurement of CSF core Alzheimer disease biomarkers for routine clinical diagnosis: do fresh vs frozen samples differ? Bellomo G, et al. Alzheimers Res Ther. 2020; 12(1): 121.
- Towards a unified protocol for handling of CSF before β-amyloid measurements. Janelidze S, et al. Alzheimers Res Ther. 2019; 11(1): 63.
Related articles
Integration of ADx NeuroSciences within Fujirebio unleashes a world of new possibilities
Fujirebio Europe N.V. will formally and fully integrate ADx NeuroSciences N.V. into its organization on April 1, 2026. This completes a process that...
Fujirebio introduces its Neuro Expert Toolbox (NExT) at AAIC 2025
Fujirebio is introducing its Neuro Expert Toolbox (NExT) for the first time at AAIC 2025 (Alzheimer's Association International Conference®) in...
Video - Alzheimer's awareness redefined
Follow the journey of the Sullivan family and leading Alzheimer’s Neurologist and Researcher Dr. David Greeley as they introduce and explain these...
Publication - Serum and cerebrospinal fluid neurofilament light chains measured by SIMOA™, Ella™, and Lumipulse™ in multiple sclerosis naïve patients
We would like to draw your attention to a first publication on our Lumipulse® G NfL solution. In this article you will find a method comparison of CSF...
CTAD 2023 – Spotlight on recent advances in blood-based biomarkers for Alzheimer’s disease
Both fluid and imaging biomarkers provide biological evidence for the underlying etiology of cognitive impairment. The core fluid biomarkers of...
IVDR status update for Fujirebio’s Neuro products
The European CE-marking is used to support registrations of in vitro diagnostic (IVD) medical devices in many jurisdictions around the world. The...
Scientific poster - Blood sample matrix validation, impact of sample freezing and method comparison analysis using the Lumipulse® G NfL blood prototype assay
This AD/PD 2023 poster investigates the agreement between matched serum and plasma samples on the Lumipulse G NfL Blood prototype assay, the impact of...
Video - A neurochemist's search to save memories
Meet Dr. Charlotte Teunissen, Professor in Neurochemistry, and her lifelong friend Christa Reinhoudt, who was diagnosed with Alzheimer's disease in...
Scientific poster - Analytical performance of the Lumipulse® G NfL CSF <RUO>
Poster presented at the AD/PD 2023
This AD/PD 2023 poster wishes to demonstrate the analytical performance of the newly developed Lumipulse G NfL CSF...
Scientific poster - CSF pTau181/Aβ1-42 ratio increases pre-analytical variability over measuring Aβ1-42 alone
In this CTAD 2022 poster, we examine the utility of CSF biomarker ratios to correct for pre-analytical variability.
Scientific poster - A fully automated and scalable plasma pTau181 assay for Alzheimer's disease
In this article, the diagnostic performance of a modified version of the Lumipulse G pTau181 CSF test is evaluated.
New criteria for Alzheimer’s disease
New criteria for different stages of AD have been suggested by the International Working Group (IWG) and the National Institute on Aging-Alzheimer’s...
Aβ deposition and clearance: a key feature of the ageing brain
This chapter looks closer at Aβ deposition and clearance as key feature of ageing brain.
Scientific poster - Reducing misdiagnosis of Alzheimer’s disease pathology utilizing CSF and amyloid PET
In this poster we examine the performance of cognitive testing alone for identification of amyloid positivity in MCI patients from the ADNI study when...
Scientific poster - Analytical performance overview of the Lumipulse® G pTau 181 Plasma RUO assay
The aim of the study, presented at the AAIC 2022, was to determine the performance of several analytical parameters, including amongst others...
Scientific poster - Analytical performance of the Lumipulse® G β-Amyloid 1-40 Plasma and Lumipulse® G β-Amyloid 1-42 Plasma RUO assays
The aim of the study, presented at the AAIC 2022, was to determine the performance of several analytical parameters, including amongst others...
CSF biochemical pattern interpretation
What are some of the best-practices of CSF biochemical pattern interpretation? In this article series we aim at highlighting the current state of...
How to perform a lumbar puncture
In this article series we aim at highlighting the current state of knowledge and the latest developments in the field of Alzheimer’s disease (AD)...
Using CSF biomarkers to link pathology and clinical presentation
In this article series we aim at highlighting the current state of knowledge and the latest developments in the field of Alzheimer’s disease (AD)...
Video - A day at the Fujirebio Neuro Center of Excellence
In this short video we show you around at the Fujirebio Neuro Center of Excellence. Right now, expectations are high for the development of blood...
Altered proteins in brain neurodegenerative diseases
In this new article series we aim at highlighting the current state of knowledge and the latest developments in the field of Alzheimer’s disease (AD)...
Scientific poster - Comparing CSF and plasma LUMIPULSE® Alzheimer’s Disease biomarker analysis to amyloid-β PET imaging
The aim of this study was to evaluate a plasma pTau biomarker as a tool for predicting amyloid pathology.
Improving clinical diagnosis of Alzheimer’s disease: Review of the available literature
In this chapter, we will review available literature on the accuracy of the underlying pathological determinations in mild cognitive impairment (MCI)...
The drawbacks of relying solely on the standard routine clinical examinations and cognitive testing
Many subtypes of disease exist under the umbrella of dementia with Alzheimer’s disease (AD) being the most common. AD-related dementia is...
Testimonial - Value of the β-Amyloid ratio and other CSF biomarkers in the Erlangen Score interpretation algorithm
By Prof. Dr. Piotr Lewczuk - Two groups of established cerebrospinal fluid (CSF) biomarkers reflect two major pathological alterations in Alzheimer's...
Scientific poster - Towards an easy plasma pTau 181 detection
Blood-based Alzheimer’s disease (AD) biomarker testing could be used as a simple, accessible, and scalable approach to help support the diagnosis of...
Webinar replay - Preparing for the future of plasma based Alzheimers disease diagnostics
At Fujirebio we are hosting a webinar series with leading expert speakers dedicated to current topics in the field of Alzheimer's disease diagnostics...
Video - The interest of automated testing for all four CSF biomarkers
In less than 2 minutes, this video explains the advantages of automated biomarker testing for all four CSF biomarkers, over other available testing...
The Fujirebio Neuro Center of Excellence
The Fujirebio Neuro Center of Excellence has been founded with the objective of rising to this challenge. It is a research and development hub focused...
Booklet - First edition of our new clinical booklet "A few drops of insight can lead to an ocean of understanding"
Early diagnosis of Alzheimer's disease is crucial. The desire to tackle neurodegenerative diseases by always finding earlier diagnostic solutions and...
Video - Ratio calculation of Aβ1-42 and Aβ1-40 offers essential information about the buildup of amyloid pathology in a patient's brain
This 2 minute video explains why and how a ratio calculation of the two amyloid proteins, Aβ1-42 and Aβ1-40, offers particularly essential information...
Video - Daniel's story about his early testing and diagnosis of Alzheimers disease
Daniel lives in Stockholm, Sweden, and was diagnosed with Alzheimer’s disease when he was still in his early 50's. In this 6-minute video we follow...
Influence of automation on Aβ1-42/Aβ1-40 ratio and its use
Automation is an important step in the direction of more standardization as it limits the number of manual handling steps and therefore minimizes...
Comparison of Aβ1-42/Aβ1-40 ratio with other ratios
CSF Aβ1-42/Aβ1-40 is a tool to normalize values of patients with different amyloid levels, as other ratios might be seen more as interpretation tools...
How to work with Aβ1-42/Aβ1-40 ratio
One cause of discordant results can be the preanalytical conditions, e.g. when laboratories use tubes that bind certain proteins. Aβ1-42 adsorption is...
Improvement of AD risk scores by use of the Aβ1-42/Aβ1-40 ratio
Different scores have been developed to provide an interpretation of biomarker results for AD diagnosis or risk prediction. Here we will give two...
Aβ1-42/Aβ1-40 ratio for interpretation of discordant results
By use of the Aβ1-42/Aβ1-40 ratio, discordant results (i.e. when amyloid and tau biomarkers are not concordant) can be improved. However, while the...
Use of the Aβ1-42/Aβ1-40 ratio to improve accuracy of AD diagnosis
It is widely agreed that, since cerebrospinal fluid (CSF) is in direct contact with the central nervous system (CNS), analytes measured in this body...
What is Alzheimer's disease?
Alzheimer’s disease, which is the most common form of dementia, is an incurable degenerative disease. Neurons in certain parts of the brain are...