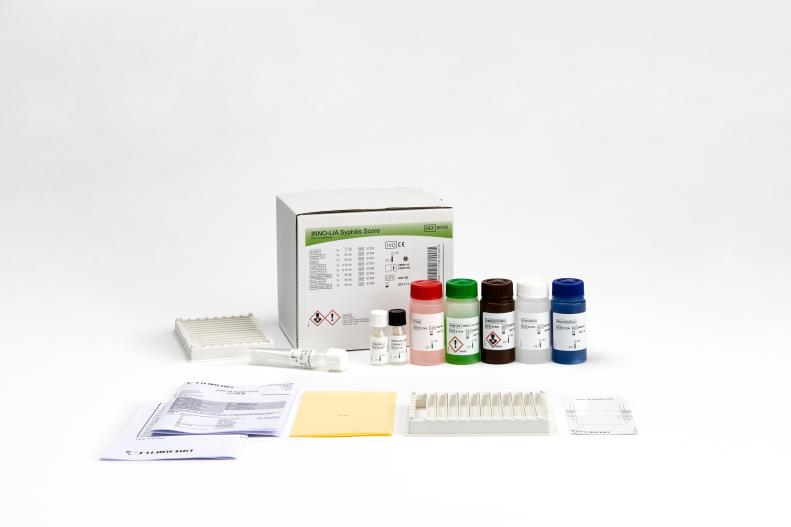
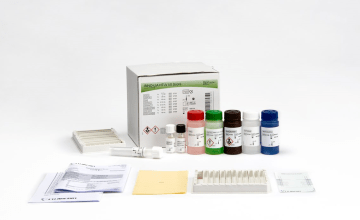
INNO-LIA® Syphilis Score
One test, one run, one final result
Line immunoassay for the confirmation of antibodies to Treponema pallidum in human serum and plasma.
Product number 80542
Click here to navigate
- Contact sales for information
- Details
- Conditions of sale
- Citations
- Documentation
- Product inquiry
- Related products
-
Details
Features & Benefits
- High sensitivity: 100% (683/683)
- Optimal specificity on clinical and potentially interfering samples: 99.5% (655/658)
- No false reactivity with potential cross-reactive pathological samples
- One assay instead of several serological assays for one confirmation result
- Color-coded reagents
- Ready-to-use reagents
- Four control lines to monitor 1) non-specific reactivity, 2) sample addition, 3) and 4) color development steps
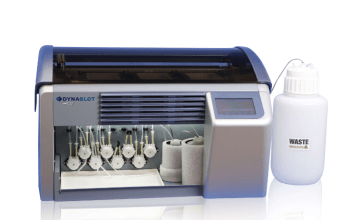
- Fully automated strip processing possible using TENDIGO™, Auto-LIA™ 48, AutoBlot 3000(H) or Roboblot®
- Objective, automated reading and interpretation of the strips possible using LiRAS® for Infectious Diseases
-
Conditions of sale
To read the end user conditions of sale for this product please visit our Resource center.
-
Citations
The BIOZ badges associated with Fujirebio products include peer-reviewed citations derived from scientific studies using Fujirebio products. Please note that the peer-reviewed citations do not reflect the regulatory status of Fujirebio products. Users should refer to the specific product documentation and any (clinical) claims made therein in order to ensure compliant use. For each country or geographic region, users must verify the related regulatory status of the Fujirebio product.
-
Documentation
Browse regulatory documents for this product
Create a (free!) eServices account and start browsing all product documentation right away.
Get access to this section and more
Create a free eServices account now and instantly access multiple digital resources:
- Product documentation
- Selected scientific posters
- Product leaflets
- How-to videos and more…
Multimedia
Watch product videos
Get a (free!) eServices account and benefit from full access to all our online resources.
-
Product inquiry
-
Related products