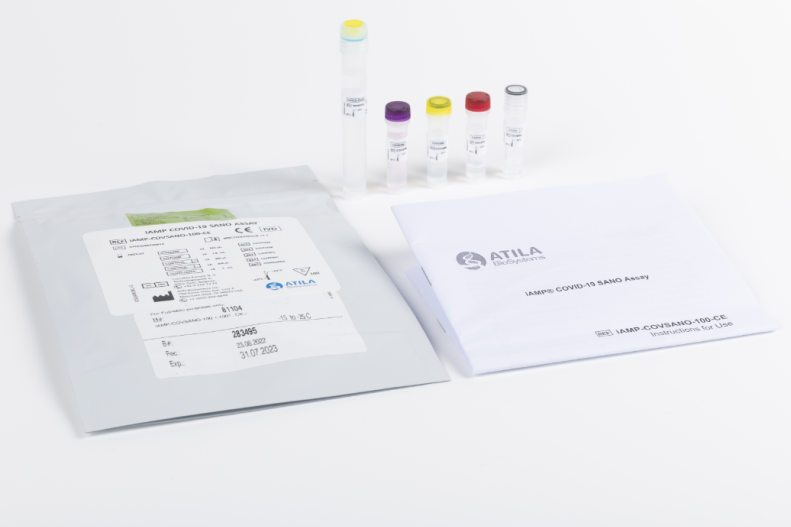
iAMP COVID-19 SANO Assay
A real-time fluorescent RT-isothermal assay based on Atila’s proprietary isothermal amplification technology intended for the qualitative detection of nucleic acid from the SARS-CoV-2 in nasopharyngeal swabs, oropharyngeal swabs and saliva specimens from individuals with signs and symptoms of infection who are suspected of COVID-19.
Fast. Simple. Accurate.
Product number 81104
Product number 80762
Product number 80812
Click here to navigate
- Details
- Conditions of sale
- Documentation
- Insights
- Related products
-
Details
Results are for the identification of SARS-CoV-2 RNA. The SARS-CoV-2 RNA is generally detectable in upper respiratory specimens during the acute phase of infection. Positive results are indicative of the presence of SARS-CoV-2 RNA; clinical correlation with patient history and other diagnostic information is necessary to determine patient’s infection status. Positive results do not rule out bacterial infection or co-infection with other viruses. Negative results do not preclude SARS-CoV-2 infection and should not be used as the sole basis for patient management decisions. Negative results must be combined with clinical observations, patient history, and epidemiological information.
The iAMP COVID-19 SANO Assay is intended for use by trained clinical laboratory personnel specifically instructed and trained in the techniques of real-time nucleic acid amplification and in vitro diagnostic procedures.
* The iAMP COVID-19 Sample Collection Device and iAMP COVID-19 Sample Collection Device NP are for Research Use Only; not for use in diagnostic procedures. Not yet available as CE marked product.
-
Conditions of sale
To read the end user conditions of sale for this product please visit our Resource center.
-
Documentation
Browse regulatory documents for this product
Create a (free!) eServices account and start browsing all product documentation right away.
Get access to this section and more
Create a free eServices account now and instantly access multiple digital resources:
- Product documentation
- Selected scientific posters
- Product leaflets
- How-to videos and more…
-
Insights
Jan 6, 2022[Jan 2022 Update] This scientific literature overview demonstrates the excellent performance of the fully automated nucleocapsid protein antigen assay Lumipulse® G SARS-CoV-2 Ag
Lumipulse G SARS-CoV-2 Ag was the first high-sensitive nucleocapsid protein antigen assay launched on a fully automated chemiluminescent platform.
I...
Aug 26, 2020Japanese authorities have chosen Lumipulse® for nasopharyngeal or saliva-based SARS-CoV-2 antigen testing of passengers at their main international airports
Several fully automated and CLEIA-based LUMIPULSE G1200 systems from Fujirebio are right now performing fast, high sensitivity SARS CoV-2 antigen...
-
Related products