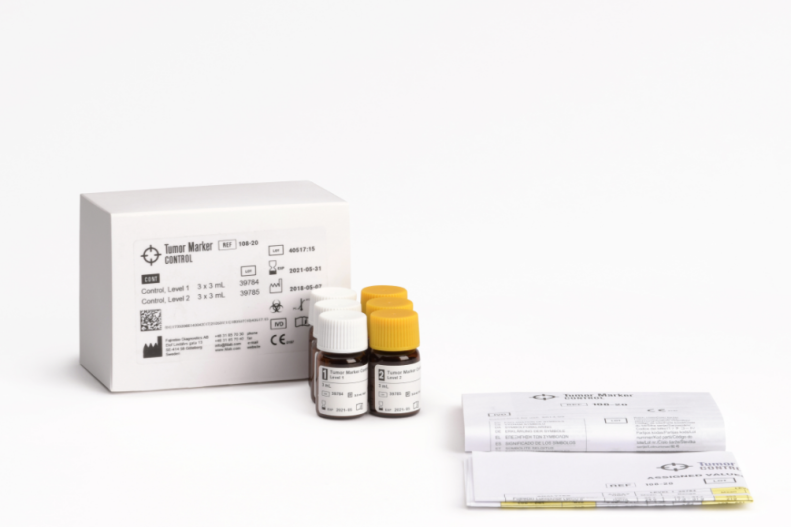
Tumor Marker Control
- The only available multi-constituent control containing the novel biomarker HE4
- Contains clinically relevant proportions of FREE PSA and PSA-ACT
- Contains medically relevant levels for HE4, AFP, CA 125, CA 15-3, CA 19-9, CEA, Ferritin, PSA, and Free PSA
- Lyophilized, 100% human serum matrix
- Reconstituted stability up to 60 days at <-20°C
- Stable for up to 9 freeze/thaw cycles
- Assayed values available for manual and automated methodologies
Product number 108-20 / 108-20W
Click here to navigate
- Contact sales for information
- Details
- Conditions of sale
- Documentation
- Product inquiry
- Related products
-
Details
Intended Use
For In Vitro Diagnostic Use Only. Fujirebio Diagnostics Tumor Marker Control is intended for use as an assayed control serum to monitor the precision of laboratory testing procedures for the analysis of AFP, CA15-3, CA19-9, CA125, CEA, Ferritin, HE4, PSA and Free PSA.
Description
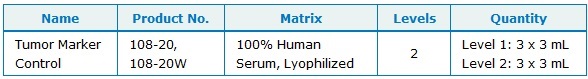
Packaging: 6 x 3 mL (3 vials of each of 2 levels – Lyophilized)
Typical Values
The values shown in the typical values below are provided solely as an example of typical values. For actual values, please refer to the lot specific assigned values sheet supplied with the product. Values were obtained from testing performed on the Abbott ARCHITECT and AxSYM Immunoassay systems except for HE4 (Fujirebio Diagnostics EIA).
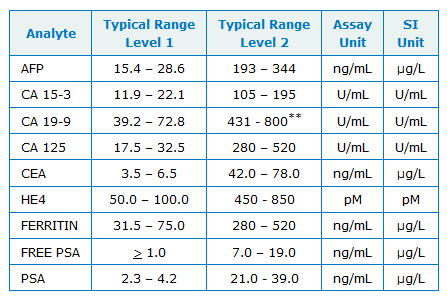
**Higher values as seen with Abbott ARCHITECT assay.
Architect and AxSYM are registered trademarks of Abbott Laboratories, Diagnostic Division, Abbott Park, IL 60064 USA
-
Conditions of sale
To read the end user conditions of sale for this product please visit our Resource center.
-
Documentation
Browse regulatory documents for this product
Create a (free!) eServices account and start browsing all product documentation right away.
Other documents
FileTumor_Marker_Control_Sheet_2010-08_r1.pdf (pdf, 1.46 mb)Get access to this section and more
Create a free eServices account now and instantly access multiple digital resources:
- Product documentation
- Selected scientific posters
- Product leaflets
- How-to videos and more…
-
Product inquiry
-
Related products