Lung Marker Control
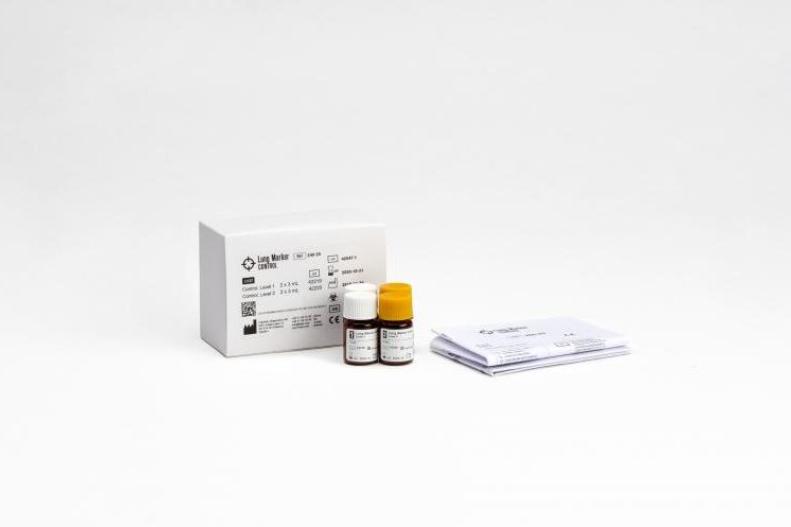
The Lung Marker Control is an assayed, two-level control containing Cyfra and ProGRP antigens.
Lung Marker Control
Product number 240-20
Level 1: 2 x 3 mL, Level 2: 2 x 3 mL
CE marked
Please contact your local Fujirebio representative for the availability of this product in your country.