New criteria for Alzheimer’s disease
In this article series we aim at highlighting the current state of knowledge and the latest developments in the field of Alzheimer’s disease (AD) testing. This chapter reviews the new criteria for different stages of AD that have been suggested by the International Working Group (IWG) and the National Institute on Aging-Alzheimer's Association (NIA-AA).
Other articles in this series:
- Altered proteins in brain neurodegenerative diseases
- Using CSF biomarkers to link pathology and clinical presentation
- How to perform a lumbar puncture
- Handling and transportation of CSF samples
- CSF biochemical pattern interpretation
- Aβ deposition and clearance: a key feature of ageing brain
New criteria for different stages of AD have been suggested by the International Working Group (IWG) and the National Institute on Aging-Alzheimer’s Association (NIA–AA) (Dubois B, et al. 2007; Albert MS, et al. 2011; McKhann GM, et al. 2011; Sperling M, et al. 2011; Dubois B, et al. 2014; Dubois B, et al. 2021).
These criteria have been constructed to permit a diagnosis of AD in earlier stages of the disease and are centered on the clinical identification of cognitive impairment subtypes together with one or more abnormal biomarkers, including MRI, PET, and CSF markers.
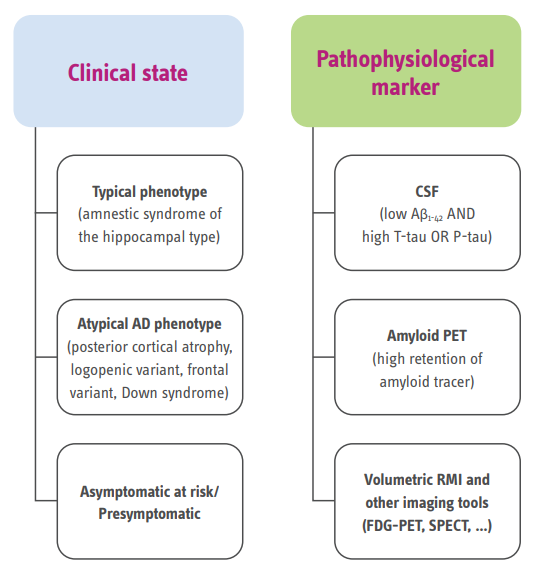
These criteria broaden the spectrum of the disease to include its preclinical states, in which Alzheimer’s pathology exists without clinical symptoms.
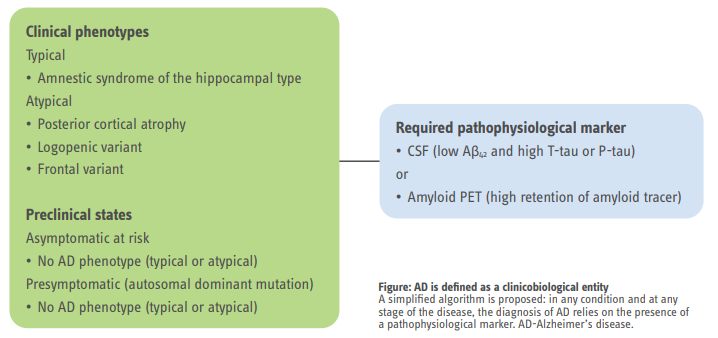
To increase diagnostic certainty, the criteria for AD incorporate biomarker evidence for pathology, which can be obtained by neuroimaging (MRI measures of atrophy, ¹⁸F-FDG PET measures of cerebral hypometabolism, and amyloid PET measures of β-amyloid deposition) and CSF testing (decreased β-amyloid concentrations and increased T-tau or P-tau concentrations).
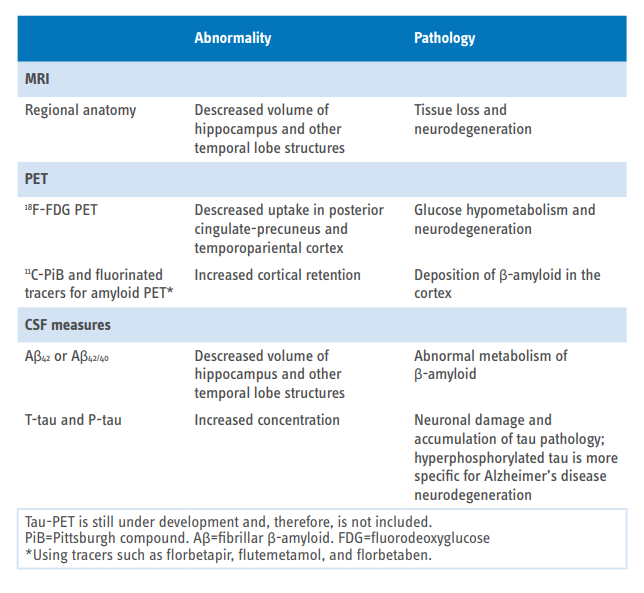
AT(N)(C) system in Alzheimer’s disease
More recently, the National Institute on Aging–Alzheimer’s Association (NIA–AA) (Jack CR et al. 2018) has proposed to define exclusively AD by its underlying neuropathologic changes that can be documented by the biomarkers in living people. The biomarker profile (pathologic process) and cognitive staging represent independent sources of information. The definition and the grading of disease severity is assessed by the biological construct of AD across its entire spectrum as a continuum.
AT(N) BIOMARKER GROUPING
- A: Aggregated Aβ amyloid peptides or associated pathologic state
(CSF Aβ42, or Aβ42/40 ratio; Amyloid PET) - T: Aggregated tau (neurofibrillary tangles) or associated pathologic state
(CSF phosphorylated tau; Tau PET) - (N): Neurodegeneration or neuronal injury
(Anatomic MRI; FDG PET; CSF total tau)
CSF VERSUS IMAGING BIOMARKERS
- The ongoing active pathologic state is denoted by CSF
- The accumulation of neuropathologic load and location in the brain is denoted by imaging
DEFINITION OF ALZHEIMER’S CONTINUUM
- A: Amyloid biomarkers (Aβ42 or Aβ42/40 ratio for CSF) determine whether or not an individual is potentially in the Alzheimer’s continuum.
- T: Pathologic tau biomarkers (P-tau for CSF) determine if someone who is in Alzheimer’s continuum has Alzheimer’s disease.
STAGING SEVERITY
- (N): Neurodegenerative/neuronal injury biomarkers (T-tau for CSF) provide powerful prediction of future cognitive decline.
- (C): Cognitive symptoms determine the syndromal categorical cognitive staging regardless the etiology.
Note: A and T are specific neuropathologic changes of AD, whereas (N) and (C) are not specific to AD and therefore placed in parentheses.
FLEXIBILITY OF THE AT(N) SYSTEM
- Each biomarker group is labeled (-) normal, (+) abnormal or (*) not determined
E.g. A+T+(N)+ to categorize AD-pathologic changes
- Alternatively to the binary approach, each biomarker group could be also labelled semi-quantitatively (0) clearly normal, (1) intermediate / marginally altered or (2) clearly abnormal.
E.g. A2T1(N)1 categorize AD profile or A2T0(N)0 if only the Amyloid biomarker is clearly abnormal
- New biomarker groups beyond AT(N) can be added when they will become available.
These guidelines would permit an important step toward harmonization of interpretation of biomarkers in a personalized medicine.
- A combination of an abnormal Aβ and a pathologic tau biomarker constitutes AD regardless of cognitive symptoms, and thus AD is a biologically defined entity.
- The staging of the severity of cognitive symptoms is independent of the underlined pathology. The syndromal categorical scheme preserves the tree clinical categories: cognitively unimpaired, mild cognitive impairment and dementia.
Bibliography
- Clinical diagnosis of Alzheimer’s disease: recommendations of the International Working Group. Dubois B, et al. Lancet Neurol. 2021; 20(6): 484-496. Review.
- NIA-AA Research Framework: Toward a biological definition of Alzheimer’s disease. Jack CR Jr, et al. Alzheimers Dement. 2018; 14(4): 535-562. Review.
- Strategic roadmap for an early diagnosis of Alzheimer’s disease based on biomarkers. Frisoni GB, et al. Lancet Neurol. 2017; 16(8): 661-676. Review.
- Are CSF Biomarkers Useful as Prognostic Indicators in Diagnostically Unresolved Cognitively Impaired Patients in a Normal Clinical Setting. Schjønning Nielsen M, et al. Dement Geriatr Cogn Dis Extra. 2016; 6(3): 465-476.
- Recommendations for cerebrospinal fluid Alzheimer’s disease biomarkers in the diagnostic evaluation of mild cognitive impairment. Herukka SK, et al. Alzheimers Dement. 2017; 13(3): 285-295. Review.